📋 Previous Year Paper
NEET 2016
Full paper · Physics, Chemistry & Biology · 180 Questions · 3 Hours 20 Minutes
180Questions
720Max Marks
200Minutes
+4 / −1Marking
💡
You're browsing as a guest. Register for free to attempt a subset of these questions and track your performance. Unlock the full paper with a one-time subscription.
📊 Your Full Test Performance Summary
NEET 2016You are yet to attempt this test. Complete the test to see your performance summary here.
180
Questions Available
0
Attempted
0
No. of Attempts
0
Correct
00:00
Total Time
00:00
Avg / Question
⚛️ Physics — 45 Questions
1
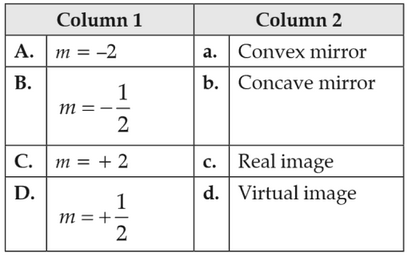
Physics+4 / −1Match the corresponding entries of Column 1 with Column 2. [Where m is the magnification produced by the mirror]

2
Physics+4 / −1What is the minimum velocity with which a body of mass m must enter a vertical loop of radius R so that can complete the loop?
3
Physics+4 / −1From a disc of radius R and mass M, a circular hole of diameter R, whose rim passes through the centre is cut. What is the moment of inertia of the remaining part of the disc about a perpendicular axis, passing through the centre?
4
Physics+4 / −1If the magnitude of sum of two vectors is equal to the magnitude of difference of the two vectors, the angle between these vectors is:
5
Physics+4 / −1When an \(\alpha\)-particle of mass m moving with velocity v bombards on a heavy nucleus of charge Ze, its distance of closest approach from the nucleus depends on m as
6
Physics+4 / −1A potentiometer wire is 100 cm long and a constant potential difference
is maintained across it. Two cells are connected in series first to
support one another and then in opposite direction. The balance points
are obtained at 50 cm and 10 cm from the positive end of the wire in the
two cases. The ratio of emf's is
7
Physics+4 / −1A particle of mass 10 g moves along a circle of radius 6.4 cm with a constant tangential acceleration. What is the magnitude of this acceleration if the kinetic energy of the particle becomes equal to 8 × 10−4J by the end of the second revolution after the beginning of the motion ?
8
Physics+4 / −1A body of mass 1 kg begins to move under the action of time dependent force \(\vec{F}\) = \(\left(2t\hat{i} + 3t^2\hat{j}\right)\) N , where \(\hat{i}\) and \(\hat{j}\) are unit vectors along x and y axis.What power will developed by the force at the time (t) ?
9
Physics+4 / −1The angle of incidence for a ray of light at a refracting surface of a prismis \(45^\circ\). The angle of prism is \(60^\circ\). If the ray suffers minimum deviation through the prism, the
angle of minimum deviation and refractive index of the material of the prism respectively, are
10
Physics+4 / −1At what height from the surface of earth the gravitation potential and the value of g are \(-\, 5.4 × 10^7J\, \text{Kg}^{−2}\) and \(6.0\,\text{ms}^{−2}\) respectively? Take the radius of earth as 6400 km.
11
Physics+4 / −1The ratio of escape velocity at earth (\(v_e\)) to the escape velocity at a planet (\(v_p\)) whose radius and mean density are twice as that of earth is
12
Physics+4 / −1A astronomical telescope has objective and eyepiece of focal lengths 40 cm and 4 cm respectively. To view an object 200 cmaway from the objective, the lenses must be separated by a distance
13
Physics+4 / −1The intensity at the maximum in a Young’s double slit experiment is \(I_0\). Distance between two slits is \(d\, =\, 5\lambda\), where \(\lambda\) is the wavelength of light used in the experiment. What will be the intensity in front of one of the slits on the screen placed at a distance D = 10 d?
14
Physics+4 / −1In a diffraction pattern due to a single slit of width a, the first minimum is observed at an angle 30° when light of wavelength 5000 Å is incident on the slit. The first secondary maximum is observed at an angle of
15
Physics+4 / −1A gas is compressed isothermally to half its initial volume. The same gas is compressed separately through an adiabatic process until its volume is again reduced to half.
16
Physics+4 / −1A refrigerator works between 4°C and 30°C. It is required to remove 600 calories of heat every second in order to keep the temperature of the refrigerated space constant.The power required is (Take, 1 cal =4.2Joules)
17
Physics+4 / −1If the velocity of a particle is \(\text{v}\, =\, \text{At}\, +\, \text{Bt}^2\), where A and B are constants, then the distance travelled by it between \(1\,s\) and \(2\,s\) is
18
Physics+4 / −1A disc and a sphere of same radius but different masses roll off on two inclined planes of the same altitude and length. Which one of the two objects gets to the bottom of the plane first?
19
Physics+4 / −1A uniform circular disc of radius 50 cm at rest is free to turn about an axis which is perpendicular to its plane and passes through its centre. It is subjected to a torque which produces a constant angular acceleration of 2.0 rad s−2. Its net acceleration in s−2 at the end of 2.0 S is approximately
20
Physics+4 / −1The molecules of a given mass of a gas have r.m.s. velocity of \(200\, \text{ms}^{−1}\) at \(27^\circ \text{C}\) and \(1.0 \times 10^5 \text{Nm}^{−2}\) pressure. When the temperature and pressure of the gas are respectively, \(127^ \circ \text{C}\) and \(0.05 \times 10^5 \text{Nm}^{ −2}\) , the rms velocity of its molecules in \(\text{ms}^{ −1}\) is
21
Physics+4 / −1An electron of mass m and a photon have same energy E. The ratio ofde-Broglie wavelengths associated with them is(c being velocity of light)
22
Physics+4 / −1When a metallic surface is illuminated with radiation of wavelength \(\lambda\), the stopping potential is \(\text{V}\). If the same surface is illuminated with radiation of wavelength \(2\lambda\), the stopping potential is \(\displaystyle \frac{V}{4}\).The threshold wavelength for the metallic surface is
23
Physics+4 / −1Two identical charged spheres suspended from a common point by two mass less strings of lengths l, are initially at a distance d (d < < l) apart because of their mutual repulsion. The charges begin to leak from both the spheres at a constant rate. As a result, the spheres approach each other witha velocity \(v\). Then, \(v\) varies as a function of the distance \(x\) between the sphere, as
24
Physics+4 / −1Coefficient of linear expansion of brass and steel rods are \(\alpha_1\) and \(\alpha_2\). Length of brass and steel rods are \(l_1\) and \(l_2\) respectively. If \(\left(l_2 − l_1\right)\) is maintained same at all temperatures, which one of the following relations holds good?
25
Physics+4 / −1A piece of ice falls from a height h so that it melts completely. Only one-quarter of the heat produced is absorbed by the ice and all energy of ice gets converted into heat during its fall. The value of \(h\) is [Latent heat of ice is \(3.4 × 10^5 J/kg\) and \(g = 10 N/kg\)
26
Physics+4 / −1A black body is at a temperature of \(5760\, \text{K}\). The energy of radiation emitted by the body at wavelength \(250\, \text{nm}\) is \(\text{U}_1\),at wavelength \(500\, \text{nm}\) is \(\text{U}_2\) and that at \(1000\, \text{nm}\) is \(\text{U}_3\) Wien's constant, \(\text{b}\, =\, 2.88 \times 10^6\, \text{nmK}\). Which of the following is correct?
27
Physics+4 / −1Two non-mixing liquids of densities \(p\) and \(np \left(n > 1\right)\) are put in a container. The height of each liquid is \(\text{"A"}\). A solid cylinder of length \(\text{L}\) and density \(\text{d}\) is put in this container. The cylinder floats with its axis vertical and length \(pL \left(p < 1\right)\) in the denser liquid. The density \(d\) is equal to
28
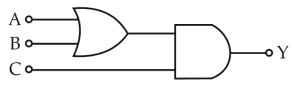
Physics+4 / −1To get output 1 for the following circuit, the correct choice for the inputis

29
Physics+4 / −1A npn transistor is connected in common emitter configuration in a given amplifier. A load resistance of \(800\, \Omega\) is connected in the collector circuit and the voltage drop across it is \(0.8\, \text{V}\). If the current amplification factor is \(0.96\) and the input resistance of the circuit is \(192\, \Omega\), the voltage gain and the power gain of the amplifier will respectively be
30
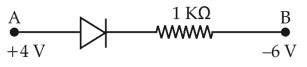
Physics+4 / −1Consider the junction diode as ideal. The value of current flowing through AB is

31
Physics+4 / −1A particle moves so that its position vector is given by \(\vec{r}\, =\, cos \,\omega \,t\, \hat{x}\, +\, sin\, \omega\,t\,\hat{y}\), where \(\omega\) is a constant. Which of the following is true ?
32
Physics+4 / −1An inductor 20 mH, a capacitor 50 μF and a resistor 40 Ω are connected in series across a source of emf
\(V\, =\, 10\,sin\,340\,t\). The power loss in A.C. circuit is
\(V\, =\, 10\,sin\,340\,t\). The power loss in A.C. circuit is
33
Physics+4 / −1A small signal voltage V (t) = V0 sin ωt is applied across an ideal capacitor C
34
Physics+4 / −1A long solenoid has 1000 turns. When a current of 4 A flows through it, the magnetic flux linked with each turn of the solenoid is 4 × 10 −3 Wb. The self-inductance of the solenoid is
35
Physics+4 / −1A siren emitting a sound of frequency \(800\, Hz\) moves away from an observer towards a cliff at a speed of \(15\, \text{ms}^{−1}\). Then, the frequency of sound that the observer hears in the echo reflected from the cliff is (Take velocity of sound in air = \(330\,\text{ms}^{−1}\))
36
Physics+4 / −1An air column, closed at one end and open at the other, resonates with a tuning fork when the smallest length of the column is \(50\, \text{Cm}\). The next larger length of the column resonating with the same tuning fork is
37
Physics+4 / −1A uniform rope of length \(L\) and mass \(m_1\) hangs vertically from a rigid support. A block of mass \(m_2\) is attached to the free end of the rope. A transverse pulse of wavelength \(λ_1\) is produced at the lower end of the rope. The wavelength of the pulse when it reaches the top of the rope is \(λ_2\).The ratio \(\displaystyle \frac{λ_1}{λ_2}\) is
38
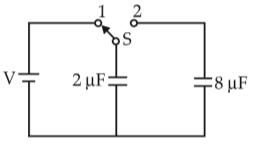
Physics+4 / −1A capacitor of 2 μF is charged as shown in the diagram. When the switch S is turned to position 2, the percentage of its stored energy dissipated is

39
Physics+4 / −1A car is negotiating a curved road of radius R. The road is banked at anangle θ. The coefficient of friction between the tyres of the car and the road is μs. The maximum safe velocity on this road is
40
Physics+4 / −1The magnetic susceptibility is negative for
41
Physics+4 / −1Given the value of Rydberg constant is \(10\,\text{m}^{-1}\), the wave number of the last line of the Balmer series in hydrogen spectrum will be
42
Physics+4 / −1The charge flowing through a resistance R varies with time t as \(\text{Q}\, =\, at − bt^2\), where a and b are positive constants. The total heat produced in R is
43
Physics+4 / −1Out of the following options which one can be used to produce a propagating electromagnetic wave?
44
Physics+4 / −1A long straight wire of radius a carries a steady current I. The current is uniformly distributed over its cross-section. The ratio of the magnetic fields B and B', at radial distances \(\displaystyle\frac{a}{2}\) and \(2a\) respectively, from the axis of the wire is
45
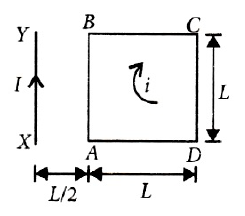
Physics+4 / −1A square loop ABCD carrying a current is placed near and coplanar with a long straight conductor XY carrying a current I, the net force on the loop will be

🧪 Chemistry — 45 Questions
46
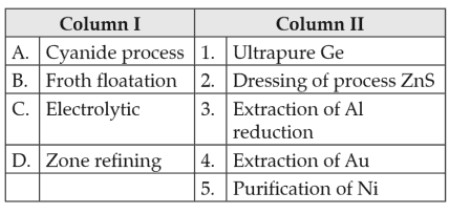
Chemistry+4 / −1Match items of Column I with the items of Column II and assign the correct code.
Codes

Codes
47
Chemistry+4 / −1Which is the correct statement for the given acids ?
48
Chemistry+4 / −1Fog is a colloidal solution of
49
Chemistry+4 / −1Which one of the following orders is correct for the bond dissociation enthalpy of halogen molecules ?
50
Chemistry+4 / −1MY and NY3, two nearly insoluble salts have the same Ksp values of 6.2 × 10-13 at room temperature. Which statement would be true in regard to MY and NY3 ?
51
Chemistry+4 / −1Which of the following statements about hydrogen is incorrect ?
52
Chemistry+4 / −1The correct statement regarding the basicity of arylamines is
53
Chemistry+4 / −1When copper is heated with conc. HNO3 it produces
54
Chemistry+4 / −1For the following reactions,
(i) CH3CH2CH2Br + KOH \(\rightarrow\) CH3CH = CH2 + KBr + H2O
(ii)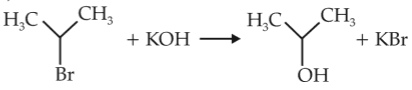
(iii)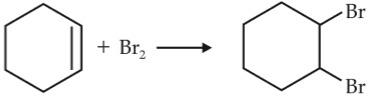
Which of the following statement is correct ?
(i) CH3CH2CH2Br + KOH \(\rightarrow\) CH3CH = CH2 + KBr + H2O
(ii)

(iii)

Which of the following statement is correct ?
55
Chemistry+4 / −1Two electrons occupying the same orbital are distinguished by
56
Chemistry+4 / −1he reaction
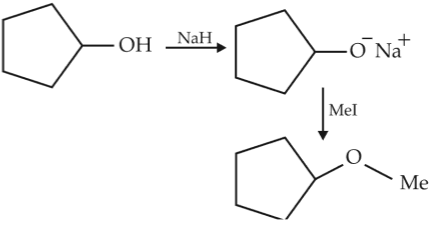
can be classified as

can be classified as
57
Chemistry+4 / −1The electronic configurations of Eu (Atomic no. 63), Gd (Atomic no. 64) and Tb (Atomic no. 65) are
58
Chemistry+4 / −1At 100°C the vapour pressure of a solution of 6.5 g of a solute in 100 g water is 732 mm. If Kb = 0.52, the boiling point of this solution will be
59
Chemistry+4 / −1The correct statement regarding the comparison of staggered and eclipsed conformations of ethane, is
60
Chemistry+4 / −1Which one of the following characteristics is associated with adsorption ?
61
Chemistry+4 / −1Match the compounds given in column I with the hybridisation and shape given in column II and mark the correct option.
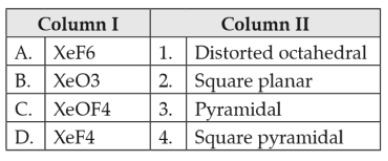

62
Chemistry+4 / −1The correct statement regarding a carbonyl compound with a hydrogen atom on its alpha-carbon, is
63
Chemistry+4 / −1In a protein molecule various amino acids are linked together by
64
Chemistry+4 / −1Which of the following is an analgesic ?
65
Chemistry+4 / −1The pair of electron in the given carbanion, CH3C≡C–, is present in which orbitals ?
66
Chemistry+4 / −1Consider the molecules CH4, NH3 and H2O. Which of the given statements is false ?
67
Chemistry+4 / −1Which one of the following statements is correct when SO2 is passed through acidified K2Cr2O7 solution ?
68
Chemistry+4 / −1 The correct thermodynamic conditions for the spontaneous reaction at all temperatures is
69
Chemistry+4 / −1Natural rubber has
70
Chemistry+4 / −1In which of the following options the order of arrangement does not agree with the variation of property indicated against it ?
71
Chemistry+4 / −1Which of the following reagents would distinguish cis - cyclopenta - 1 , 2 - diol from the trans - isomer ?
72
Chemistry+4 / −1The product obtained as a result of a reaction of nitrogen with CaC2 is
73
Chemistry+4 / −1Equal moles of hydrogen and oxygen gases are placed in container with a pin - hole through which both can escape . What fraction of the oxygen escapes in the time required for one - half of the hydrogen to escape ?
74
Chemistry+4 / −1Lithium has a bcc structure . Its density is 530 kg m-3 and its atomic mass is 6.94 g mol-1 . Calculate the edge length of a unit cell of lithium metal.
( NA = 6.02 × 1023 mol-1 ).
( NA = 6.02 × 1023 mol-1 ).
75
Chemistry+4 / −1 Which of the following statements about the composition of the vapour over an ideal 1 : 1 molar mixture of benzene and toluene is correct ? Assume that the temperature is constant at 25 ° C.
( Given , vapour pressure data at 25 ° C , benzene = 12.8 kPa , toluene = 3.85 kPa )
( Given , vapour pressure data at 25 ° C , benzene = 12.8 kPa , toluene = 3.85 kPa )
76
Chemistry+4 / −1 Which of the following has longest C – O bond length ? ( Free C - O bond length in CO is 1.128 Å . )
77
Chemistry+4 / −1Among the following , the correct order of acidity is
78
Chemistry+4 / −1 In the reaction,
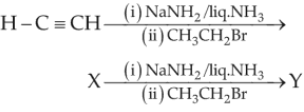
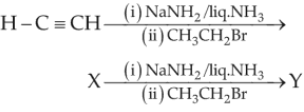
79
Chemistry+4 / −1Consider the nitration of benzene using mixed conc . H2SO4 and HNO3 . If a large amount of KHSO4 is added to the mixture , the rate of nitration will be
80
Chemistry+4 / −1 The product formed by the reaction of an aldehyde with a primary amine is
81
Chemistry+4 / −1The pressure ofH2 required to make the potential of H<2/sub> - electrode zero in pure water at 298 K is
82
Chemistry+4 / −1The correct statement regarding RNA and DNA , respectively is
83
Chemistry+4 / −1Which one given below is a non - reducing sugar ?
84
Chemistry+4 / −1Consider the following liquid - vapour equilibrium
Liquid \(\rightleftharpoons\) Vapour
Which of the following relations is correct ?
Liquid \(\rightleftharpoons\) Vapour
Which of the following relations is correct ?
85
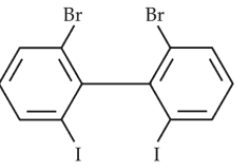
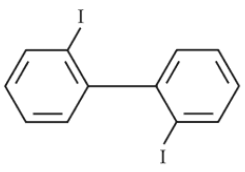
Chemistry+4 / −1Which of the following biphenyls is optically active ?
86
Chemistry+4 / −1Which of the following statements is false ?
87
Chemistry+4 / −1The ionic radii of A + and B- ions are 0.98 × 10-10 m and 1.81 × 10-10 m . The coordination number of each ion in AB is
88
Chemistry+4 / −1The rate of a first - order reaction is 0.04 mol L-1S-1 at 10 sec and 0.03 mol L-1S-1 at 20 sec after initiation of the reaction . The half - life period of the reaction is
89
Chemistry+4 / −1The addition of a catalyst during a chemical reaction alters which of the following quantities ?
90
Chemistry+4 / −1Predict the correct order among the following .
🧬 Biology — 90 Questions
91
Biology+4 / −1Mitochondria and chloroplast are
I. semi-autonomous organelles
II. formed by the division of pre existing organelles and they contain DNA but lack protein synthesising machinery
Which one of the following options is correct?
I. semi-autonomous organelles
II. formed by the division of pre existing organelles and they contain DNA but lack protein synthesising machinery
Which one of the following options is correct?
92
Biology+4 / −1Chrysophytes, euglenoids, dinoflagellates and slime moulds are included in the kingdom
93
Biology+4 / −1A tall true breeding garden pea plant is crossed with a dwarf true breeding garden pea plant. When the F1 plants were selfed, the resulting genotypes were in the ratio of
94
Biology+4 / −1Depletion of which gas in the atmosphere can lead to an increased incidence of skin cancers
95
Biology+4 / −1In the stomach, gastric acid is secreted by the
96
Biology+4 / −1Which of following is wrongly matched in the given table ?
97
Biology+4 / −1Which of the following is not a stem modification ?
98
Biology+4 / −1Select the incorrect statement
99
Biology+4 / −1One of the major components of cell wall of most fungi is
100
Biology+4 / −1Which of the following statements is not correct ?
101
Biology+4 / −1Which of the following is not a characteristic feature during mitosis in somatic cells ?
102
Biology+4 / −1In context of amniocentesis, which of the following statement is incorrect ?
103
Biology+4 / −1The Taq polymerase enzyme is obtained from
104
Biology+4 / −1The standard petal of a papilionaceous corolla is also called
105
Biology+4 / −1Which of the following characteristic features always holds true for the corresponding group of animals ?
106
Biology+4 / −1Changes in GnRH pulse frequency in females is controlled by circulating levels of
107
Biology+4 / −1Microtubules are the constituents of
108
Biology+4 / −1Photosensitive compound in human eye is made up of
109
Biology+4 / −1The primitive prokaryotes responsible for the production of biogas from the dung of ruminant animals, include the
110
Biology+4 / −1Identify the correct statement on ‘inhibin’.
111
Biology+4 / −1It is much easier for a small animal to run uphill than for a large animal, because
112
Biology+4 / −1Which one of the following is a characteristic feature of cropland ecosystem ?
113
Biology+4 / −1Tricarpellary, syncarpous gynoecium is found in the flowers of
114
Biology+4 / −1In which of the following, all the three are macronutrients ?
115
Biology+4 / −1Reduction in pH of blood will
116
Biology+4 / −1Lack of relaxation between successive stimuli in sustained muscle contraction is known as
117
Biology+4 / −1Which one of the following statements is wrong ?
118
Biology+4 / −1Which of the following is a restriction endonuclease?
119
Biology+4 / −1Which of the following would appear as the pioneer organisms on bare rocks ?
120
Biology+4 / −1Water vapour comes out from the plant leaf through the stomatal opening. Through the same stomatal opening carbon dioxide diffuses into the plant during photosynthesis. Reason out the above statements using the following options.
121
Biology+4 / −1Cotyledon of maize grain is called
122
Biology+4 / −1Which of the following guards the opening of hepatopancreatic duct into the duodenum ?
123
Biology+4 / −1 In the mammals, which blood vessel would normally carry largest amount of urea ?
124
Biology+4 / −1The term ecosystem was coined by
125
Biology+4 / −1Which of the following is required as inducer(s) for the expression of lac operon ?
126
Biology+4 / −1When does the growth rate of a population following the logistic modal equals zero ? The logistic model is given as
\(\displaystyle \frac{\text{dN}}{\text{dt}}\) = \(\displaystyle \text{rN} \left(\frac{\text{1-N}}{\text{K}}\right)\)
\(\displaystyle \frac{\text{dN}}{\text{dt}}\) = \(\displaystyle \text{rN} \left(\frac{\text{1-N}}{\text{K}}\right)\)
127
Biology+4 / −1Which one of the following statements is not true ?
128
Biology+4 / −1In bryophytes and pteridophytes, transport of male gametes requires
129
Biology+4 / −1Which one of the following cell organelles is enclosed by a single membrane?
130
Biology+4 / −1Analogous structures are a result of
131
Biology+4 / −1Which one of the following statements is wrong ?
132
Biology+4 / −1 Proximal end of the filament of stamen is attached to the
133
Biology+4 / −1Which of the following is not required for any of the techniques of DNA fingerprinting available at present ?
134
Biology+4 / −1Which one of the following characteristics is not shared by birds and mammals ?
135
Biology+4 / −1The amino acid, tryptophan is the precursor for the synthesis of
136
Biology+4 / −1Joint Forest Management Concept was introduced in India during
137
Biology+4 / −1A complex of ribosomes attached to a single strand of RNA is known as
138
Biology+4 / −1Which of the following features is not present in the phylum - Arthropoda?
139
Biology+4 / −1Asthma may be attributed to
140
Biology+4 / −1Pick out the correct statements
I. Haemophilia is a sex linked recessive disease
II . Down's syndrome is due to aneuploidy
III . Phenylketonuria is an autosomal recessive gene disorder
IV . Sickle cell anaemia is an X linked recessive gene disorder
I. Haemophilia is a sex linked recessive disease
II . Down's syndrome is due to aneuploidy
III . Phenylketonuria is an autosomal recessive gene disorder
IV . Sickle cell anaemia is an X linked recessive gene disorder
141
Biology+4 / −1The two polypeptides of human insulin are linked together by
142
Biology+4 / −1The coconut water from tender coconut represents
143
Biology+4 / −1Which of the following is not a feature of the plasmids ?
144
Biology+4 / −1Which is the National Aquatic Animal of India ?
145
Biology+4 / −1The Avena curvature is used for bioassay of
146
Biology+4 / −1Which of the following is the most important cause of animals and plants being driven to extinction?
147
Biology+4 / −1Which of the following approaches does not give the defined action of contraceptive ?
Microbe: Product
Microbe: Product
148
Biology+4 / −1In a test cross involving F1 dihybrid flies, more parental-type offspring were produced than the recombinant type offspring. This indicates
149
Biology+4 / −1A typical fat molecule is made up of
150
Biology+4 / −1Match the terms in Column I with their description in Column II and choose the correct option.
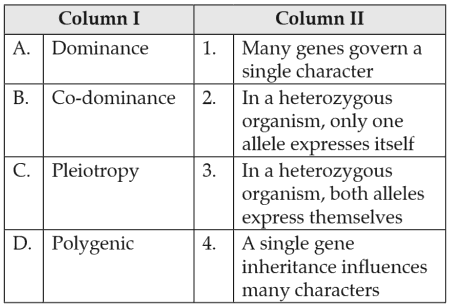

151
Biology+4 / −1Which of the following features is not present in Periplaneta americana ?
152
Biology+4 / −1Water soluble pigments found in plant cell vacuoles are
153
Biology+4 / −1A cell at telophase stage is observed by a student in a plant brought from the field. He tells his teacher that this cell is not like other cells at telophase stage. There is no formation of cell plate and thus the cell is containing more number of chromosomes as compared to other dividing cells. This would result in
154
Biology+4 / −1A plant in your garden avoids photorespiratory losses, has improved water use efficiency, shows high rates of photosynthesis at high temperatures and has improved efficiency of nitrogen utilisation. In which of the following physiological groups would you assign this plant ?
155
Biology+4 / −1In higher vertebrates, the immune system can distinguish self-cells and non-self. If this property is lost due to genetic abnormality and it attacks self-cells, then it leads to
156
Biology+4 / −1Emerson's enhancement effect and red drop have been instrumental in the discovery of
157
Biology+4 / −1Select the correct statement
158
Biology+4 / −1Blood pressure in the pulmonary artery is
159
Biology+4 / −1Which of the following structures is homologous to the wing of a bird ?
160
Biology+4 / −1Seed formation without fertilisation in flowering plants involves the process of
161
Biology+4 / −1Name the chronic respiratory disorder caused mainly by cigarette smoking
162
Biology+4 / −1Spindle fibres attach on to
163
Biology+4 / −1Stems modified into flat green organs performing the functions of leaves are known as
164
Biology+4 / −1In a chloroplast the highest number of protons are found in
165
Biology+4 / −1Nomenclature is governed by certain universal rules. Which one of the following is contrary to the rules of nomenclature ?
166
Biology+4 / −1In meiosis crossing over is initiated at
167
Biology+4 / −1Antivenom injection contains preformed antibodies while polio drops that are administered into the body contain
168
Biology+4 / −1Which of the following most appropriately describes haemophilia ?
169
Biology+4 / −1Which part of the tobacco plant is infected by Meloidogyne incognita ?
170
Biology+4 / −1Which of the following statements is wrong for viroids ?
171
Biology+4 / −1Which of the following statements is not true for cancer cells in relation to mutations ?
172
Biology+4 / −1Which type of tissue correctly matches with its location ?
173
Biology+4 / −1Which of the following pairs of hormones are not antagonistic (having opposite effects) to each other ?
174
Biology+4 / −1Specialised epidermal cells surrounding the guard cells are called
175
Biology+4 / −1Fertilisation in humans is practically feasible only if
176
Biology+4 / −1Which one of the following is the start codon ?
177
Biology+4 / −1A river with an inflow of domestic sewage rich in organic waste may result in
178
Biology+4 / −1Following are the two statements regarding the origin of life
I. The earliest organisms that appeared on the earth were non-green and presumably anaerobes
II. The first autotrophic organisms were the chemoautotrophs that never released oxygen
Of the above statements which one of the following options is correct ?
I. The earliest organisms that appeared on the earth were non-green and presumably anaerobes
II. The first autotrophic organisms were the chemoautotrophs that never released oxygen
Of the above statements which one of the following options is correct ?
179
Biology+4 / −1A system of rotating crops with legume or grass pasture to improve soil structure and fertility is called
180
Biology+4 / −1Gause’s principle of competitive exclusion states that